ABOUT SYMBIO, YOUR NICHE CRO
Symbio is a client-focused, full-service contract research organization (CRO) specializing in dermatology, aesthetics, gastroenterology, and inflammatory diseases.
As your niche CRO, we offer flexible study delivery backed by deep therapeutic expertise and responsive teams to bring products and devices to market faster.

How Symbio Became the Largest Specialty CRO
In November of 2023, Symbio, LLC and Dow Development Laboratories, LLC merged with Proinnovera to create Symbio Proinnovera, one of the world’s largest dermatology-focused clinical research organizations.
In September of 2024, the group was renamed Symbio and began further expanding into dermatology-related therapeutic areas based on the clinical translation, our client’s needs, and our team and KOL’s expertise in these broadening areas.
Today, we are one of the top dermatology CROs and specialists in aesthetics, gastroenterology, and inflammatory diseases, offering:
- A strong site network for global reach and rapid enrollment
- Access to best-in-class industry KOLs
- De-risked execution thanks to a specialized team and effective vendor oversight
- Deep experience from 650+ studies in 85+ indications across dermatology and related therapeutic areas
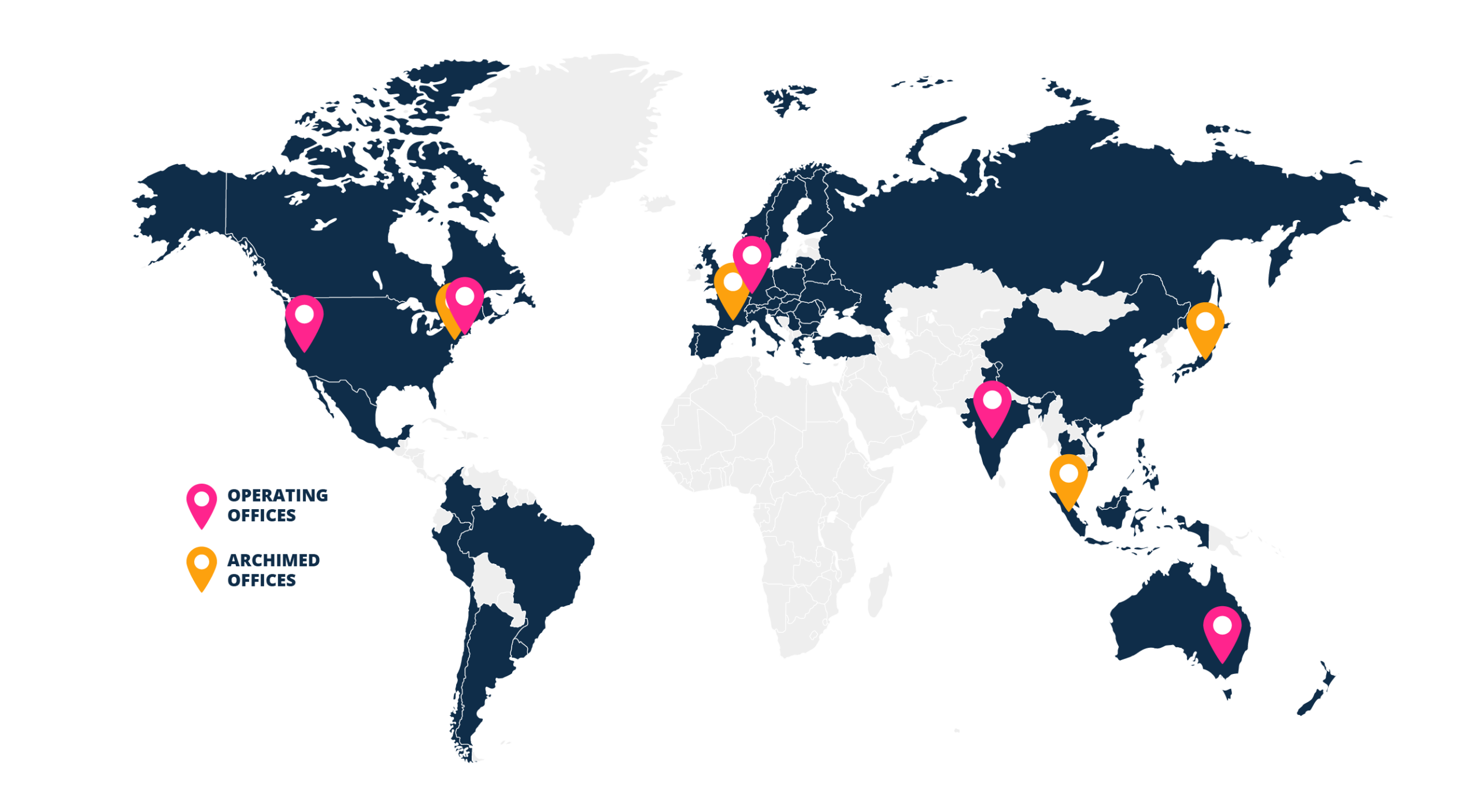
International Footprint
Headquartered in the USA with offices across Europe and in Australia and subsidiaries around the globe, we offer optimal team alignment in 50 countries and counting.
The Symbio Site Network
Developing and maintaining excellent relationships with experienced clinical research sites is vital to the success of any study and every CRO. It’s especially important in dermatology research, aesthetics, gastroenterology, and inflammation research, where unique patient populations, study, designs, and regulatory demands make sites even more integral to trial success.
As a specialized CRO, we take great pride in the strong relationships we have established with specialized sites and investigators over the years. With our high-performing sites by our side, we streamline recruitment and deliver shorter enrollment timelines.


A Message From Symbio’s CEO
At Symbio, our clients are the core of everything we do. We recognize the significant responsibility of bringing drugs and devices to market, and we are truly honored to be chosen as a partner to help guide this critical journey.
With a vast network of experts across specialized therapeutic areas and global regions, we provide customized solutions tailored to meet the unique needs of each client.
Our reputation is built on the principle that “the reward for good work is more work.” As your specialty CRO and committed partner, we remain dedicated to delivering exceptional service every step of the way.
— Alan Morgan



